Below are some PDFs that Spartan Mosquito of Hattiesburg, Mississippi, shared with state pesticide officials and the Environmental Protection Agency to get registrations for its two products, the Spartan Mosquito Eradicator and the Spartan Mosquito Pro Tech (EPA #93813-1). I am sharing the files here so that regulatory officials who have not seen the data can better evaluate whether the company’s claims are supported. Additionally, I am hoping that mosquito biologists might read the documents and leave comments on whether the experimental protocols are scientifically sound. To facilitate these goals I’ve listed some draft notes under each file.
The efficacy claims made by Spartan Mosquito
The Spartan Mosquito Eradicator (ingredients: sugar, yeast, and sodium chloride) is advertised to eliminate up to 95% of mosquitoes for up to 90 days. The Spartan Mosquito Pro Tech (ingredients: sugar, yeast, boric acid), registered by the EPA in 2020, is advertised to “kill mosquitoes” for up to 30 days. The company also claims that both of the devices are so attractive to mosquitoes that they will “gather” around the tubes, crawl through small holes (11/64″, 4.4 mm) in the caps, then ingest the fluid inside.
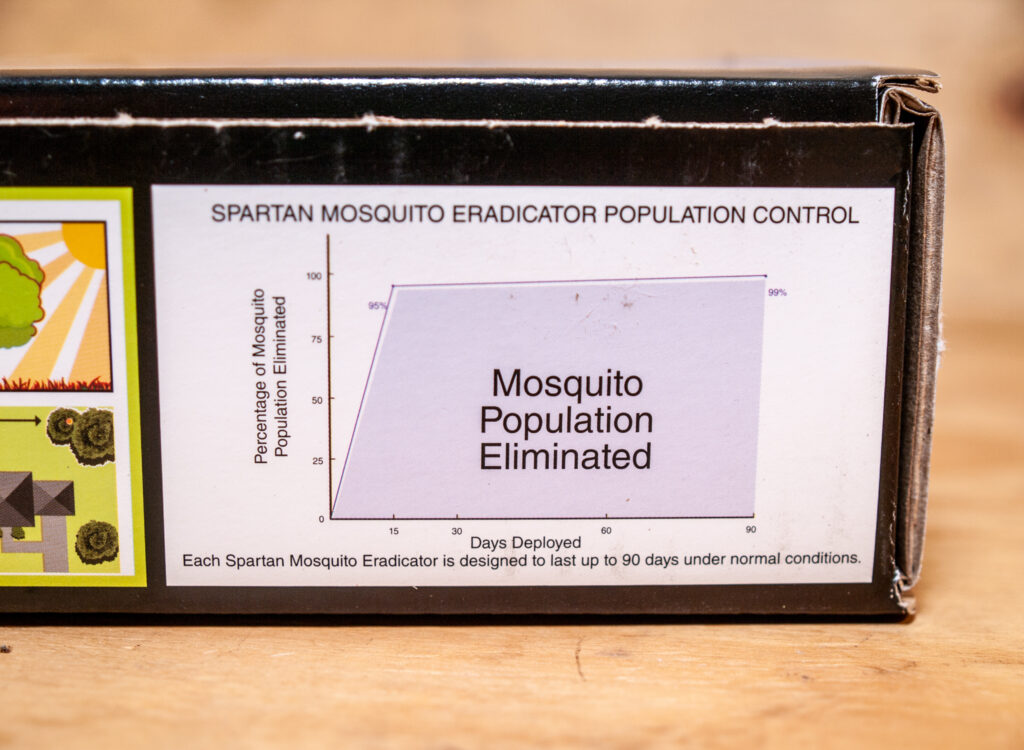
Spartan Mosquito Eradicator field efficacy data
The PDF below contains mosquito count data (number landing on forearm in 1-min interval) at two locations (New Augusta and Hattiesburg) in Mississippi during 2016. These data were sent to states that required efficacy data prior to the registration of the Spartan Mosquito Eradicator.
Notes about the above file:
- “Control” sites did not have tubes filled with water, sugar, and yeast and thus were not true controls.
- In at least one of the two studies the “test” area was situated at a residence while the “control” area was set up in a wooded area. That could bias the results. Here are details:
- The coordinates given for the “test” and “control” sites for New Augusta studies are identical (31°16’39.04″N 89° 3’37.88″W). The study street address is listed as 458 Buck Creek Road, New Augusta, MS but that does not exist; it could be 458 Buck Creek Road, Richton, MS, about 1.5 miles away. It is thus unclear where the “test” and “control” sites are actually located.
- The test site at 217 Dover Trace, Hattiesburg, was Jeremy Hirsch’s yard. He situated the control in a wooded area approximately 2300 feet north of his house. The City of Hattiesburg as well as Forrest County regularly treats residential areas with insecticide to keep mosquito populations low.
- All data collection, analysis, and writing was done by Jeremy Hirsch, a co-founder of Spartan Mosquito, so potential conflict of interest (COI).
- The regulatory consultant, Micah T. Reynolds (Technology Sciences Group, Inc.), states in the document, “This study was not conducted in full compliance with Good Laboratory Practices as outlined in 40 CFR 160.”
Spartan Mosquito Pro Tech data
The subheadings below all relate to the Spartan Mosquito Pro Tech, the version that contains sugar, yeast, boric acid. The PDFs contain efficacy data from mosquitoes inside cages, efficacy data from field trials, and a document (a “bridging rationale”) that argues Anopheles mosquitoes will be killed by the tubes as well.
Laboratory efficacy
This PDF describes mortality experiments using mosquitoes inside cages.
Notes about the above file:
- According to the procedure described, the tubes with boric acid seem to lack the yeast that is present in the version on the market.
- It is unclear whether the “control” tubes (sugar water only) had caps (like the “test” tubes did). If they did not they cannot be viewed as controls.
- There was no replication.
- Michael S. Bonner is the father of Chris Bonner, the Vice-President of Spartan Mosquito (potential COI).
- Bonner Analytical Testing Company is where Chris Bonner works (potential COI).
- There is no mention of who collected the data and whether they were qualified or employed by Spartan Mosquito (potential COI).
- The regulatory consultant, Micah T. Reynolds (Technology Sciences Group, Inc.), states in the document, “This study was not conducted in full compliance with Good Laboratory Practices as outlined in 40 CFR 160.”
The PDF below contains additional mortality experiments using mosquitoes inside aquariums and BugDorms.
Notes about the above file:
- There’s no mention of yeast being used in either test or control tubes so the conclusions would not apply to the commercial version that includes sugar, boric acid, and yeast.
- Control tubes lack caps so they cannot be viewed as acceptable controls.
- Michael S. Bonner is the father of Spartan Mosquito’s Vice President (potential COI).
- Chris Bonner works at Bonner Analytical Testing Company (potential COI).
- There is no mention of who collected the data and whether they were qualified or employed by Spartan Mosquito (potential COI).
- The regulatory consultant, Micah T. Reynolds (Technology Sciences Group, Inc.), states in the document, “This study was not conducted in full compliance with Good Laboratory Practices as outlined in 40 CFR 160.”
Field efficacy
This file includes mosquito count data collected at four test locations (and controls) in Mississippi using CDC Light Traps baited with Spartan Mosquito tubes filled with sugar, yeast, and water (for CO2 production, apparently). The PDF also contains information on the company’s standard operating procedures for field trials.
Notes about the above file:
- It is unclear whether control control tubes were used at control sites and also what ingredients they contained.
- In the Standard Operating Procedures, mosquito counts were determined using a CDC Light Trap equipped with a Spartan Mosquito tube as source of carbon dioxide. This is unusual. Most mosquito biologists would use dry ice or compressed CO2. Per the Office of the Indiana State Chemist, Jeremy Hirsch said that the Spartan Mosquito tubes do not generate enough CO2 to attract mosquitoes.
- The selection of sites to use for “test” and “control” treatments does not appear to be random. In general, it appears that control sites were situated in wooded areas and the test sites in residential areas. These decisions might influence whether sites were sprayed during town and county mosquito-abatement activities. Sites might also differ in mosquito density and species composition. The location details are presented below:
- In project BA041317, test site 1 is at edge of plowed field and near paved road, test site 2 (28 Country Rd) is at a home, and the control is in woods near a pond.
- In project BA071917, the test site was at 33 Franklin Place, a home in suburban development (across the street from Maxwell Bonner, relative of Michael and Chris Bonner and also employed by Bonner Analytical Testing Company). The control site was situated at the edge of a cemetery near a wooded area, far from the road. I.e., non-random site choice.
- In project BA072017, test site was 2711 Oak Grove Road, a residence next to Bonner Analytical Testing Company, on a busy road. It was/is owned by the Bonners. The control site was the same as in above (cemetery near woods). I.e., non-random site choice.
- In project BT0813018-03, test site given as 207 LaSalle Court but no such address exists. The true location is likely 307 LaSalle Court, a home owned by a J. Tatum, a relative of Josephine Tatum Hood-Hirsch (Spartan Mosquito’s treasurer and wife of Jeremy Hirsch). The control site was situated in “vacant wooded parcel“. I.e., non-random site choice.
- Studies were conducted by Jeremy Hirsch (then the President of Spartan Mosquito), Chris Bonner (Vice President), or Michael S. Bonner. I believe they collected data alone. Potential COIs.
- The regulatory consultant, Micah T. Reynolds (Technology Sciences Group, Inc.), states in the document, “This study was not conducted in full compliance with Good Laboratory Practices as outlined in 40 CFR 160.”
Notes about the above file:
- If Standard Operating Procedures were followed to obtain counts, company used Spartan Mosquito tubes to generate carbon dioxide for the CDC Light Traps. There is no evidence that Spartan Mosquito’s tubes generate sufficient CO2 to attract mosquitoes.
- Jeremy Hirsch and Chris Bonner are co-founders (potential COI), and Michael S. Bonner is the father of Chris Bonner (potential COI).
- There is no mention of who collected the data and whether they were qualified or employed by Spartan Mosquito.
- The regulatory consultant, Micah T. Reynolds (Technology Sciences Group, Inc.), states in the document, “This study was not conducted in full compliance with Good Laboratory Practices as outlined in 40 CFR 160.”
Literature on attractive toxic sugar baits
The PDF below is a summary of select scientific articles that Spartan Mosquito believes are supportive of the ability of the Spartan Mosquito Pro Tech to kill mosquitoes. In particular, the document argues that Anopheles species would be susceptible to attractive toxic sugar baits that contain boric acid.
Notes about the above file:
- The literature appended in the original document can be downloaded in this PDF (56MB).
- In the “Product Background” (page 5), it is stated that the inert ingredients (plural) attract the mosquitoes. There is literature cited that supports sugar being an attractant, but not yeast (the other inactive ingredient). Yeast is not mentioned anywhere in the document, including in all the appended scientific articles.
- The “Product Background” states that cap has openings that allow mosquitoes to enter but there is no cited literature on how many mosquitoes will enter holes that size when they are in search of sugar and/or yeast. Document does not mention the grid size of the Westham ATSB registered by the EPA (Micah T. Reynolds was involved in that product’s registration, too).
- “Carbon dioxide” appears 27 times and “CO2” appears 35 times in the appended articles but is not mentioned in the text of “bridging rational” authored by the regulatory consultant. It is unclear why the articles on carbon dioxide are appended. Perhaps to imply that the tubes emit sufficient quantities of carbon dioxide to entice mosquitoes inside?
- There is no literature cited that relates to whether mosquitoes enter small holes that are emitting carbon dioxide. There are several articles on holes in mosquito netting that could have been cited but they do not deal with holes this small.
- There is no literature presented that discuss whether Anopheles mosquitoes respond to sugar, yeast, and carbon dioxide differently than Aedes and Culex.
- There is no literature presented that discusses whether Anopheles will enter small holes.
Third party efficacy data
In addition to the in-house data, Spartan Mosquito also payed scientists to evaluate the tubes in their respective research laboratories at universities. It’s my understanding that these data showed the tubes were not effective, but I haven’t been able to see the data and I don’t know which versions (the Eradicator or the Pro Tech) were tested. I suspect the data have not been seen by state and federal pesticide regulators. I’ve been told that the scientists were required to sign non-disclosure agreements so the data will likely only be released if a criminal investigation somehow removes the NDAs.
There are, however, two peer-reviewed journal articles that directly relate to Spartan Mosquito’s tubes. The first (Aryaprema et al. 2020) was a direct test of Spartan Mosquito Eradicators worked (they did not) and the second (Yee et al. 2020) evaluated whether ingestion of salt water kills adult mosquitoes (it does not).
The only third-party test of the Spartan Mosquito Pro Tech was one that I conducted. There have been no peer-reviewed studies, however.
Who regulates the Spartan Mosquito Eradicator?
Because the scientific publications above reveal that the efficacy claims for the Spartan Mosquito Eradicator are false and misleading, the device does not qualify for exemption under FIFRA 25(b), the category that includes pesticides with “low risk” active ingredients such as table salt. Therefore, the Spartan Mosquito Eradicator cannot legally be sold unless it gets a registration from the EPA. Some states have already denied registrations for this very reason, but the majority seem to be allowing the illegal sales to continue. As just one example, Florida still allows sales even though state law prohibits any false or misleading claims to be made on pesticide packaging, plus prohibits sale of pesticides that have been shown under laboratory conditions to be ineffective. The latter legislation is particularly relevant given that the laboratory publishing the journal article above (Aryaprema et al. 2020) is based in Florida.
The EPA, similarly, seems to be allowing sales of the Spartan Mosquito Eradicator to continue even though the device does not have an EPA registration number. I’m not aware of other pesticides that have enjoyed immunity from both state and federal regulatory authority. It will be interesting to see how long this immunity lasts.
For more information, please see my other posts about Spartan Mosquito.